- Atoms Are Made Up Of Mostly
- What Are Atoms Made Of
- Atoms Are Made Up Of What Particles
- Atoms Are Made Up Of Space
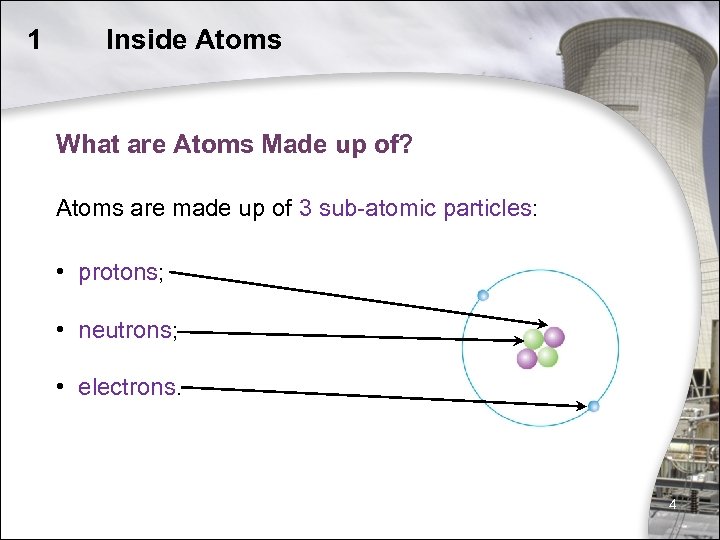
An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud. The electrons carry a negative charge and the protons carry a positive charge. Atoms are made up of three subatomic particles known as electrons (carry negative charge),protons (carry positive charge)and nuetrons (carry no charge). Protons and nuetrons together constitute nucleus which is about 1/100,000 of size of whole atom.
Background: Atoms and Light Energy
Atoms and Light Energy
| The study of atoms and their characteristics overlap several differentsciences. Chemists, Physicists, and Astronomers all must understand themicroscopic scale at which much of the Universe functions in order to seethe 'bigger picture'. |
Inside the Atom
| The ground state of an electron, the energy level itnormally occupies, is the state of lowest energy for that electron. |
| There is also a maximum energy that each electron canhave and still be part of its atom. Beyond that energy, the electronis no longer bound to the nucleus of the atom and it is considered tobe ionized. |
| When an electron temporarily occupies an energy state greater than itsground state, it is in an excited state.An electron can become excited if it is given extra energy, such as if itabsorbs a photon, or packet of light, or collides with a nearby atom or particle. |
Light Energy
Atoms Are Made Up Of Mostly
Each orbital has a specific energy associated with it. For an electronto be boosted to an orbital with a higher energy, it must overcome thedifference in energy between the orbital it is in, andthe orbital to which it is going. This means that it must absorb a photonthat contains precisely that amount of energy, or take exactly that amountof energy from another particle in a collision.The illustrations on this page are simplified versions of real atoms,of course. Real atoms, even a relatively simple ones like hydrogen,have many different orbitals, and so there are many possible energieswith different initial and final states. When an atom is in an excitedstate, the electron can drop all the way to the ground state in one go,or stop on the way in an intermediate level.
| Electrons do not stay in excited states for very long - they soonreturn to their ground states, emitting a photon with the sameenergy as the one that was absorbed. |
Identifying Individual Types of Atoms

Transitions among the various orbitals are unique for each element because theenergy levels are uniquely determined by the protons and neutrons in thenucleus. We know that different elements have different numbers ofprotons and neutrons in their nuclei. Whenthe electrons of a certain atom return to lower orbitalsfrom excited states, the photons they emit have energies that arecharacteristic of that kind of atom. This gives eachelement a unique fingerprint, making it possible to identify the elementspresent in a container of gas, or even a star.
We can use tools like the periodic table of elements to figure out exactlyhow many protons, and thus electrons, an atom has. First of all, we knowthat for an atom to have a neutral charge, it must have the same number ofprotons and electrons. If an atom loses or gains electrons, it becomesionized, or charged. The periodic table will give us the atomic number ofan element. The atomic number tells us how many protons an atomhas. For example, hydrogen has an atomic number of one - which means it hasone proton, and thus one electron - and actually has no neutrons.
What Are Atoms Made Of
For the StudentBased on the previous description of the atom, draw amodel of the hydrogen atom. The 'standard' model of an atom is knownas the Bohr model. |
Different forms of the same chemical element that differ only by the numberof neutrons in their nucleus are called isotopes. Most elements havemore than one naturally occurring isotope. Many more isotopes have beenproduced in nuclear reactors and scientific laboratories. Isotopes usuallyaren't very stable, and they tend to undergo radioactive decay untilsomething that is more stable is formed. You may be familiar with the elementuranium - it has several unstable isotopes, U-235 being one of the mostcommonly known. The 235 means that this form of uranium has 235 neutronsand protons combined. If we looked up uranium's atomic number, and substractedthat from 235, we could calculate the number of neutrons that isotope has.
Here's another example - carbon usually occurs in the form of C-12 (carbon-12), that is, 6 protons and 6 neutrons, though one isotope is C-13, with 6protons and 7 neutrons.
For the StudentUse the periodic table and the names of the elements given below to figure out how many protons, neutrons and electrons they have.Draw a model of an atom of the following element: silicon-28,magnesium-24, sulphur-32, oxygen-16, and helium-4. |
For the StudentUsing the text, define the following terms: energy levels, absorption, emission, excited state, ground state, ionization,atom, element, atomic mass, atomic number, isotope. |
A Optional Note on the Quantum Mechanical Nature of AtomsWhile the Bohr atom described above is a nice wayto learn about the structure of atoms, it is not the most accurate way tomodel them. Although each orbital does have a precise energy, theelectron is now envisioned as being smeared out in an 'electron cloud'surrounding the nucleus. It is common to speak of the mean distance to thecloud as the radius of the electron's orbit.So just remember, we'll keep the words 'orbit' and 'orbital', though we arenow using them to describe not a flat orbital plane, but aregion where an electron has a probability of being. Electrons are kept near thenucleus by the electric attraction between the nucleus and the electrons.Kept there in the same way that the nine planets stay near the Sun instead ofroaming the galaxy. Unlike the solar system, where all the planets' orbitsare on the same plane, electrons orbits are more three-dimensional. Eachenergy level on an atom has a different shape. There are mathematicalequations which will tell you the probability of the electron's locationwithin that orbit. Let's consider thehydrogen atom, which we already drew a Bohr model of.
The text and images in this section were adapted from Dave Slaven's page onThe Atom (see References below). |
Reference URLs:
The Atom
http://webs.morningside.edu/slaven/Physics/atom/
Spectra
http://www.colorado.edu/physics/PhysicsInitiative/Physics2000/quantumzone/
The Periodic Table
http://www.webelements.com/
Atoms Are Made Up Of What Particles
 Back to the Main Spectra Unit Menu
Back to the Main Spectra Unit MenuAtoms Are Made Up Of Space
Familiar and 'ordinary' matter, like the wood in a desk, the meat on your bones, the stones in your garden, as well as the
Atoms make up elements
Hydrogen is what we call an element. Iron, carbon, oxygen, sulphur and uranium are all elements. There are currently
their atoms collapse and decay almost as soon as you detect them. Fortunately, however, enough are stable to give the 92
000 000 atoms of iron (1 x 10^25 or 1E+25 atoms).
Atoms make molecules
Atoms can stick or bond together in certain permissible combinations to form groups of atoms called molecules. In the
as what you might find in a rock shop. These rings, and other similar groups of atoms, are called molecules. Atoms of different
of atom. Water is made up of H2O molecules - molecules made up of two hydrogen (H) atoms and one oxygen (O) atom
Atoms are made up of vibrations or waves
Think of a water wave on the sea. You would be right in thinking that a wave is a repeating or periodic oscillation or vibration,
vibrations. Atoms are no exception, so we need to say a little about waves first. Consider a simple wave, either a water wave
